Unit Cell Chemistry Simple Cubic, Body Centered Cubic, Face Centered Cubic Crystal Lattice Structu - YouTube
What is Atomic Packing Factor (and How to Calculate it for SC, BCC, FCC, and HCP)? – Materials Science & Engineering

Chemistry - Liquids and Solids (27 of 59) Crystal Structure: Density of the Unit Cell: Body Centered - YouTube

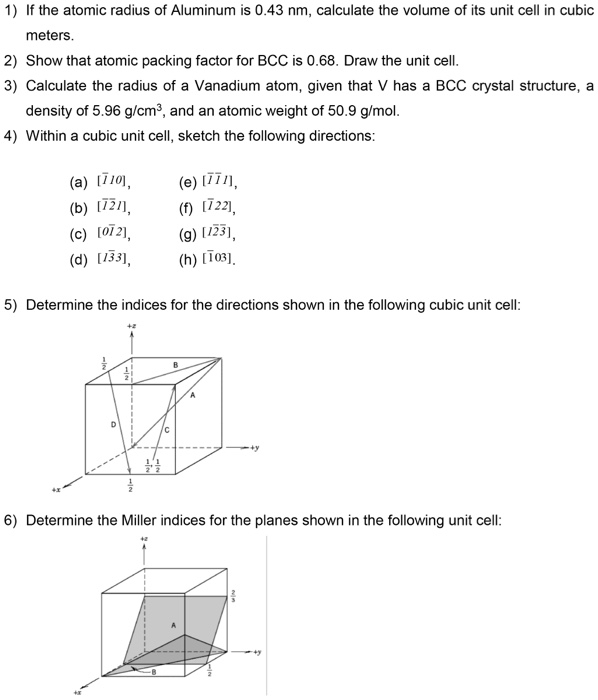
Calculate the number of unit cells in 8.1 g of aluminium if it crystallizes in a face - centred cubic (f.c.c.) structure? (Atomic mass of Al = 27 g mol^-1)